HSA halts sale of 'weight-loss' products containing high levels of banned substance
-
By A Mystery Man Writer
-
-
4.9(406)

Product Description
The “Anyang Herbal Red” weight-loss product that was found to contain high levels of sibutramine. The Health Sciences Authority (HSA) has issued a public alert on the Anyang Herbal Blue and Anyang Herbal Red weight-loss products, which have been found to contain high levels of a banned substance called sibutramine. Anyang Herbal Red was also found to contain undeclared potent Western medicines, namely diclofenac (a painkiller) and phenolphthalein (a laxative), said the HSA in a release on Monday (6 March)..
The “Anyang Herbal Red” weight-loss product that was found to contain high levels of sibutramine. The Health Sciences Authority (HSA) has issued a public

HSA issues alert on 5 cosmetic creams with very high levels of mercury and potent undeclared ingredients, Singapore News - AsiaOne

IJERPH August-1 2019 - Browse Articles

HSA warns against consuming 2 slimming products with banned substance

TRU NIAGEN - Patented Nicotinamide Riboside NAD+ Supplement. NR Supports Cellular Energy Metabolism & Repair, Vitality, Healthy Aging of Heart, Brain & Muscle - 30 Servings / 30 Capsules - Pack of 1 : Health & Household

HSA warns public to stay away from 2 weight loss products sold online

Banned substance, potent adulterants found in five products, including slimming pills: HSA

Federal Register :: Patient Protection and Affordable Care Act, HHS Notice of Benefit and Payment Parameters for 2024
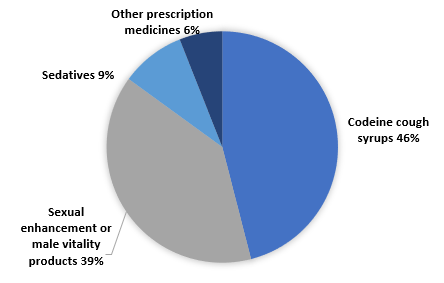
HSA Seized Over 737,000 Units of Illegal Health Products Worth $640,000 in 2022 Through Concerted Enforcement Efforts

Public Notification: A1 Slim contains hidden drug ingredients

HSA issues warning for 3 products containing banned weight loss medicine, steroids - CNA

Stratum Nutrition Nutraceuticals World
















